Modern food traceability software for food businesses

Key takeaways
The US is improving food safety by enforcing traceability in the food supply chain
Food businesses were originally expected to comply with FSMA 204 by 2026, but now there is some breathing room until 2028
Compliance is required for many items, including those on the FDA Food Traceability List (FTL), ranging from leafy greens to nut butters and finfish
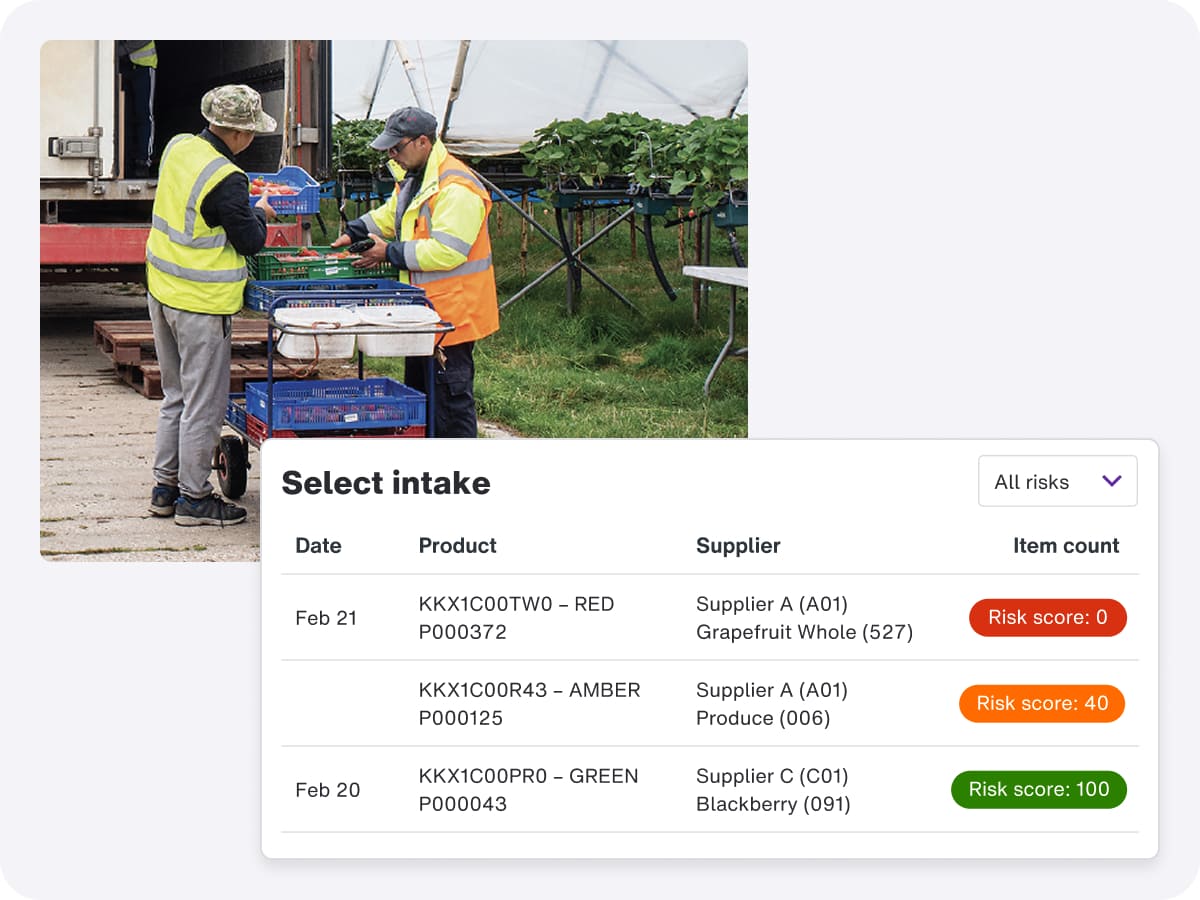
Food traceability software that can help your business comply with FSMA 204
Executive summary
The US is improving food safety by enhancing traceability in the food supply chain. As a result, food businesses need to improve their traceability capabilities and digitize their recordkeeping so they can easily share critical data with the FDA, enhance audit defensibility and data accuracy, and bolster their recall readiness.
If you’re reorganizing your operations to adopt suitable US food traceability software tools that will ensure compliance, choosing the right software can be a challenge. We’ve outlined what’s essential to look for in a solution and how TELUS Food Safety & Quality can meet all your needs.
From audits to efficiency: What US food businesses need from modern traceability software
The US is making moves to improve food safety by enhancing traceability in the food supply chain. Because of how food gets from farm to table, much of the onus of implementing the FDA’s new traceability system in the food industry will rest on the shoulders of food businesses like yours. If you still work on paper or in disconnected spreadsheets and you have disorganized recordkeeping practices, you’ll need to dramatically enhance and digitize your systems in order to meet new standards and improve audit defensibility, data accuracy and recall readiness.
Why food businesses are reassessing traceability tools
You’re likely looking to adopt a US food traceability software option that can help you meet the FDA’s new requirements and comply with FSMA 204 (The Food Traceability Final Rule, section 204 of the Food Safety Modernization Act). The FDA introduced FSMA 204 to enhance traceability in the food industry and enhance their ability to easily identify and remove contaminated food from the market. The original FSMA 204 compliance date was January 20, 2026, but many businesses weren’t ready for it so the FDA extended it to July 20, 2028, giving you more time to get up to speed.
The deadline extension may feel like a relief, but there’s still work to do. You know FSMA 204 is coming, but what needs to change now? You now have the breathing room to reassess your traceability tools and food safety programs so you can determine what needs to change within your operations. You know you need new software, but which software solution is the right one for your operations? Ultimately the tool you choose should support your organization beyond compliance.
The shift in expectations: Beyond FSMA readiness
Compliance for FSMA 204 isn’t straightforward. It’s not simply adding a step in a tried and true process. Your business may need to undertake an operations and recordkeeping overhaul. If you’re transitioning from paper and disconnected spreadsheet recordkeeping methods, implementing radically new systems and processes may feel overwhelming.
The new tools that your business adopts will need to support you and your team beyond FSMA compliance, enabling record digitization and enhanced searchability. The software you adopt will also need to work across the supply chain, not just in silos, as the records will need to make their way through the chain as needed, when needed. If the FDA wants to track a dangerous food product that’s working its way down the line, you’ll need to be able to share critical data with them in a short timeframe and so will your supply chain partners. You’ll also be expected to be able to share critical data with those partners should the FDA knock on their door.
Core requirements for modern traceability software
When it comes to selecting suitable traceability software, there are a two core capabilities that businesses must prioritize:
FSMA 204 compliance
Flexible data capture and record digitization
Support for FSMA 204 (CTEs, KDEs, 24-hour records)
Your business will be audited by the FDA after the 2028 compliance deadline to ensure that your recordkeeping systems are compliant. Suitable traceability software options that facilitate FSMA 204 compliance will be able to:
Capture Critical Tracking Events (CTEs)
What's entailed
CTEs are defined points in the food supply chain where traceability data can be collected. They mark a significant movement or change to a food product such as:
Harvesting
Cooling
Transformation
The benefits
CTEs help you closely track hazardous food products. Capturing them means no more guessing where a batch went sideways.
Record Key Data Elements (KDEs)
What's entailed
KDEs record important details related to a product's CTEs. They’re the "who, what, where and when" details that must be recorded for food products as they move through the supply chain.
The benefit
When linked together, KDEs instantly paint a complete picture of a product's journey.
Compile and share 24-hour records
What's entailed
You’ll need to be able to pull an electronic, sortable spreadsheet of traceability data for the FDA within 24 hours of a request. These records help the FDA trace the origin and presence of hazardous food products in the supply chain in a timely fashion.
The benefit
Turns a 2-day panic into a 2-minute "Export" button.
Your compliance and data will help your business mitigate risk and enable the FDA to identify the source of a foodborne illness outbreak, including managing and preventing further contamination. The economy and food businesses like yours stand to benefit greatly from this new system because by pinpointing the exact batch of contaminated food, it can be more easily extracted from the market, avoiding category-wide panic where consumers stop buying all of your product. Instead, you can isolate the affected lot of lettuce heads for example and surgically remove them, potentially avoiding huge losses and damage to your brand’s reputation.
Flexible data capture and digitization
A suitable tool needs to enable flexible data capture across different operations and easy digitization capabilities for manual and spreadsheet based processes. It should also create and store searchable, electronic records that are easily retrievable and shareable on demand.
Operational requirements that often get overlooked
Before you run out to buy the first software solution you see that mentions FSMA compliance, certain operational requirements should be heavily factored in when you select an option. Your goal should be to choose a tool that maximizes usability with minimum adaptation. The right tool should:
Pose minimal disruption to your existing workflows
Be easy to use for your frontline and seasonal staff
Scale as your volumes and partners increase
Integrate with your existing systems and partners
You need the right tool for your operations, so talk to service providers before taking a leap, to avoid purchasing a mismatch.
Turning compliance data into operational value
High quality software can help with so much more than just FSMA 204 compliance. It can enhance your operations as a whole. The right solution will help you enable:
Faster recall readiness: Isolate a single lot in seconds, not days.
Frontline simplicity: Easy-to-use tools and simplified reporting that your team can master.
Total visibility: Good data leads to better decision making , less waste and reduced risk.
Time savings: Smart digitization reduces manual work and leads to fewer errors and less data duplication.
Enhanced sharing and data storage: Strengthen your partnerships, compliance and communication.
What success looks like for US food businesses
With the right FSMA compliance solution, you'll be able to provide compliant records; instill digitized, standardized traceability processes within your operations; undertake audits confidently; and enjoy calmer recalls.
The right digital solution will help you:
Easily generate FDA-compliant spreadsheets
Enhance and streamline reporting and recordkeeping
Protect and preserve your data and make data transfer easier
Automate reporting to comply with the 24-hour rule
Record real-time, accurate traceability data
Easily track down and share that data across the supply chain
Uncover key strategies for proactive food safety management
Download the whitepaper
How TELUS supports US food businesses
We know that choosing digital tools for FSMA compliance can feel daunting, but there’s no need to fret about FSMA. TELUS Food Safety & Quality ticks all the boxes, meeting all your FSMA compliance needs while seamlessly integrating into your existing operations and dramatically enhancing your digitization capabilities. Using it will save you valuable time, reduce errors, mitigate risk, increase visibility and strengthen your partnerships and communication. Beyond ensuring compliance with the right software, TELUS is a partner you can count on to guide and support you as you enhance your operations and grow your business.